Bones & Joints Care
Orthopedics and Joint Replacement Department
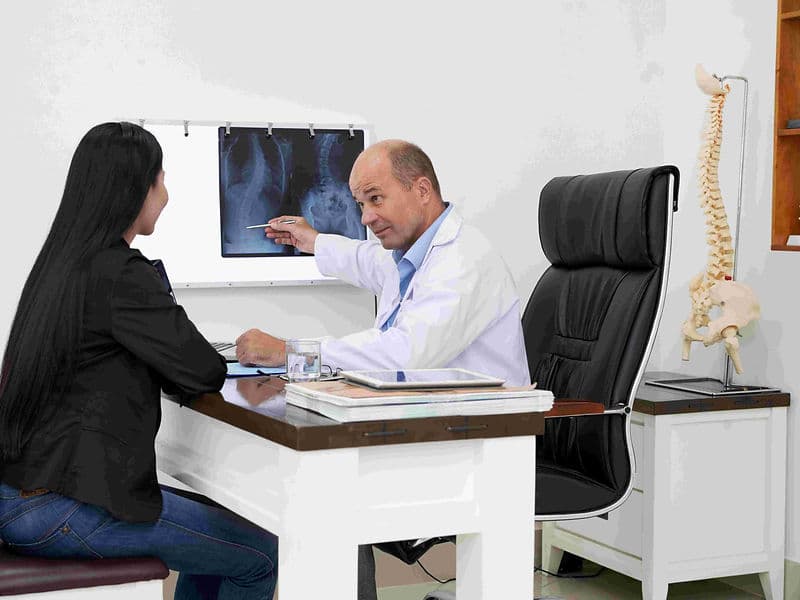
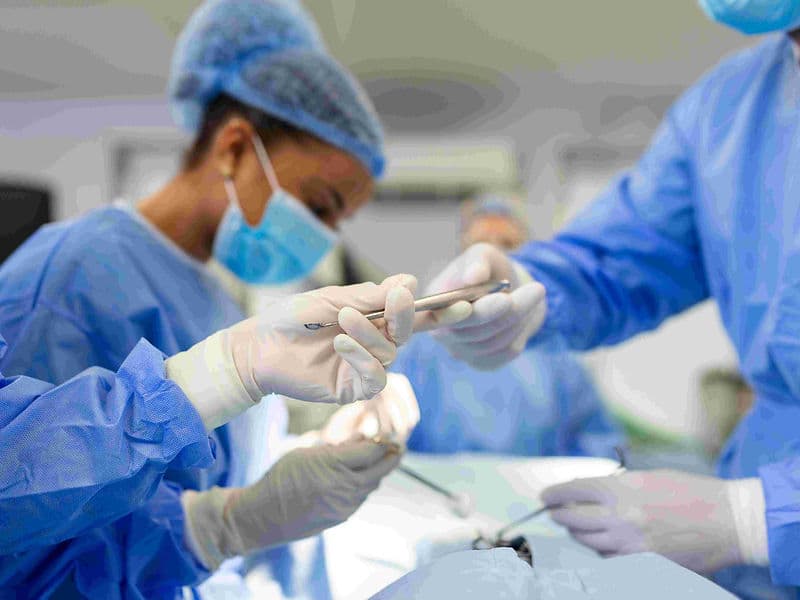
The Orthopedics and Joint Replacement Department is a center of surgical excellence where high-end engineering meets biological repair. This department focuses on restoring structural integrity and mobility, managing everything from acute sports injuries to chronic degenerative conditions like arthritis. For international patients, it provides a world-class destination for precision-driven bone and joint care.
Sub-Specialized Expert Teams
To ensure the highest clinical standards, the department is divided into focused expert units:
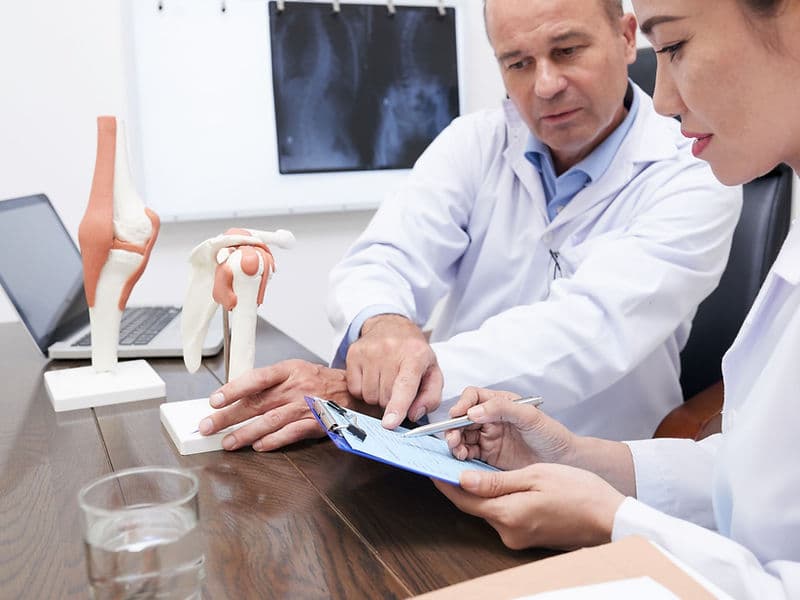
Joint Replacement Surgeons: Specialists in "resurfacing" worn-out joints through Total Knee (TKR) and Total Hip (THR) replacements.
Trauma & Fracture Surgeons: Experts in repairing complex injuries using high-grade internal fixation hardware like plates and screws.
Sports Medicine Specialists: Dedicated to "keyhole" repairs of soft tissues, including ACL reconstructions and rotator cuff repairs.
Spine Surgeons: Specialists treating the vertebral column for conditions like slipped discs, sciatica, and scoliosis.
Rheumatologists: Medical experts who manage autoimmune joint diseases such as Rheumatoid Arthritis.
Advanced Technology and Infrastructure
The department utilizes cutting-edge surgical tools to ensure sub-millimeter precision:
Robotic Navigation Systems: GPS-like sensors that assist surgeons in making perfect bone cuts for longer-lasting implants.
High-Definition Arthroscopy: Miniature cameras that allow for major joint repairs through tiny incisions, speeding up recovery.
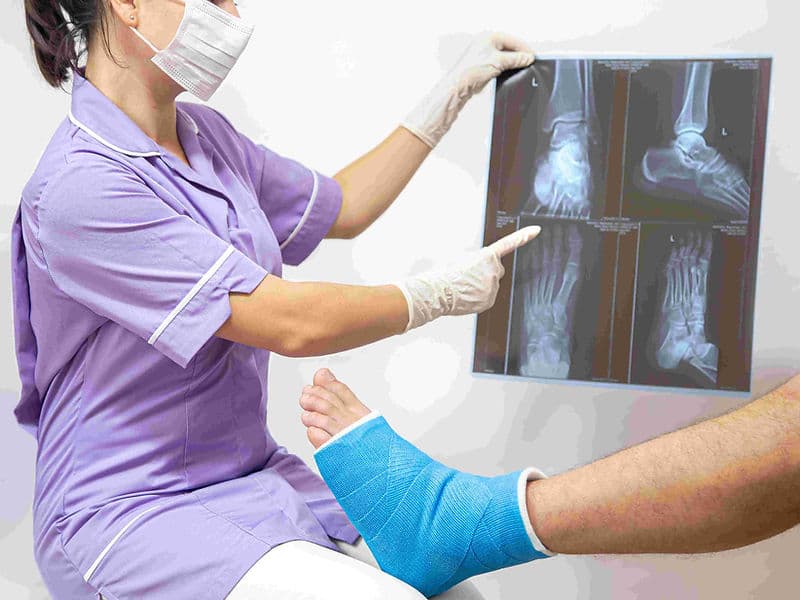
Mobile C-Arm Imaging: Real-time X-ray feedback during surgery for accurate placement of screws and implants.
Bio-Compatible Hardware: Next-generation joint replacements crafted from high-durability Titanium, Ceramic, and Cobalt-Chrome.
Comprehensive Diagnostic & Recovery Facilities
Patient care is supported by dedicated on-site units to ensure a seamless experience:
Imaging & Densitometry Lab: Features 3D CT reconstruction, high-tesla MRI, and DEXA scans for bone density assessment.
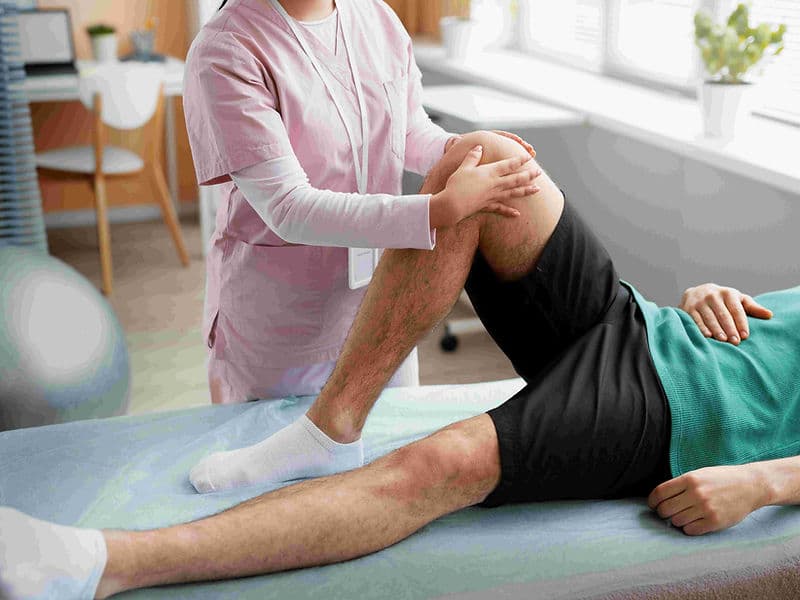
Integrated Physiotherapy Wing: A specialized rehab center focused on immediate mobilization and regaining range of motion.
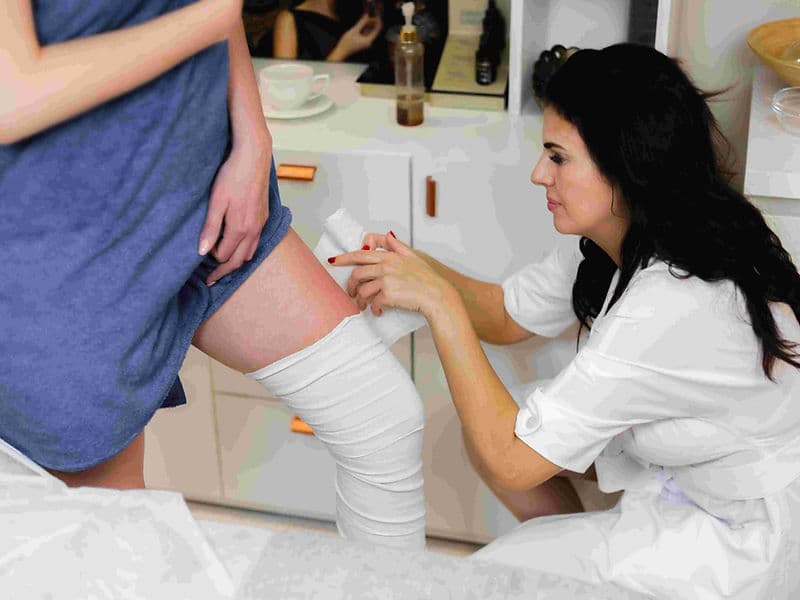
Digital Cast Room: For the application of lightweight, modern fiberglass casts for non-surgical fracture management.
International Service Suite: A professional space dedicated to managing the travel, insurance, and medical records of global patients.