Heart Care
Heart Care Department (Cardiac Sciences)
The Heart Care Department, often categorized under Cardiac Sciences, is a world-class center dedicated to the body’s most vital engine. This specialized wing is divided into two powerhouse disciplines: Cardiology (medical and minimally invasive care) and Cardiac Surgery (advanced surgical intervention). For international patients, this department offers a seamless "Heart Team" approach, addressing everything from the heart’s complex "plumbing" (blood flow) to its delicate "electricity" (rhythmic signals) with elite global standards.
A Specialized "Heart Team" of Experts
Patient care is managed by a synchronized group of specialists who focus on different dimensions of cardiac health:
Interventional Cardiologists: Experts in "keyhole" vascular repair who use advanced stents to open blocked arteries through the wrist or groin.
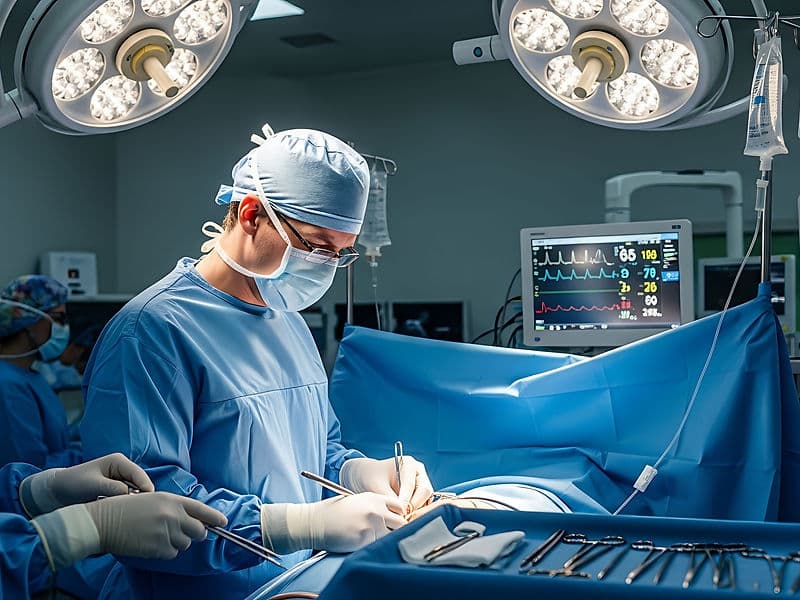
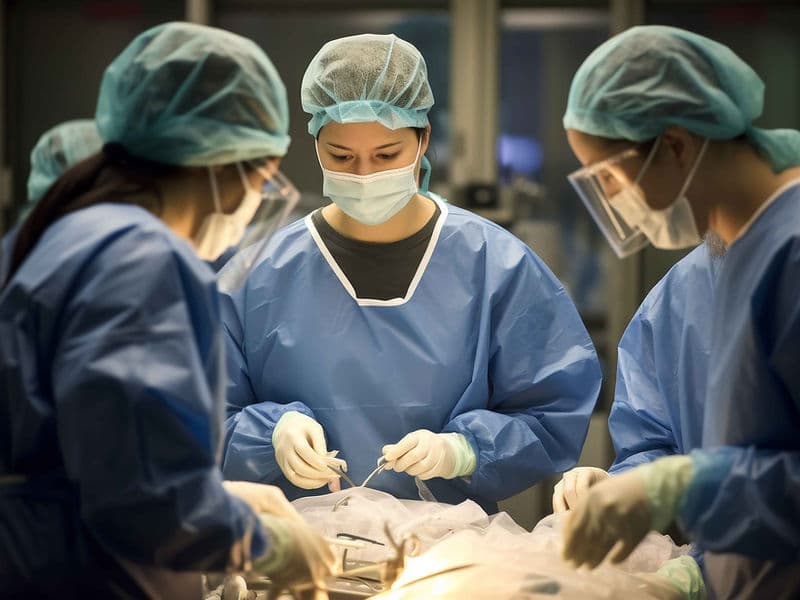
Cardiothoracic Surgeons (CTVS): The high-precision surgical team performing complex Bypass (CABG) and open-heart valve replacements.
Cardiac Electrophysiologists: The "electricians" of the heart who treat irregular rhythms (arrhythmias) and implant life-saving pacemakers or ICDs.
Non-Invasive Cardiologists: Diagnostic masters who utilize advanced external imaging like 3D Echocardiograms to map heart function.
Heart Failure Specialists: Focused on advanced management, including mechanical heart pumps (LVADs) and transplant coordination.
Cardiac Rehabilitators: Dedicated therapists who guide patients through structured recovery to strengthen the heart post-procedure.
Advanced Infrastructure and Life-Support Technology
The department functions as a high-tech "command center," utilizing the most sophisticated equipment available in modern medicine:
The Digital Cath Lab: A specialized suite for real-time vascular mapping and emergency angioplasty to resolve heart attacks instantly.
Hybrid Operating Theaters: Futuristic suites that allow for simultaneous surgical and catheter-based procedures on the same patient.
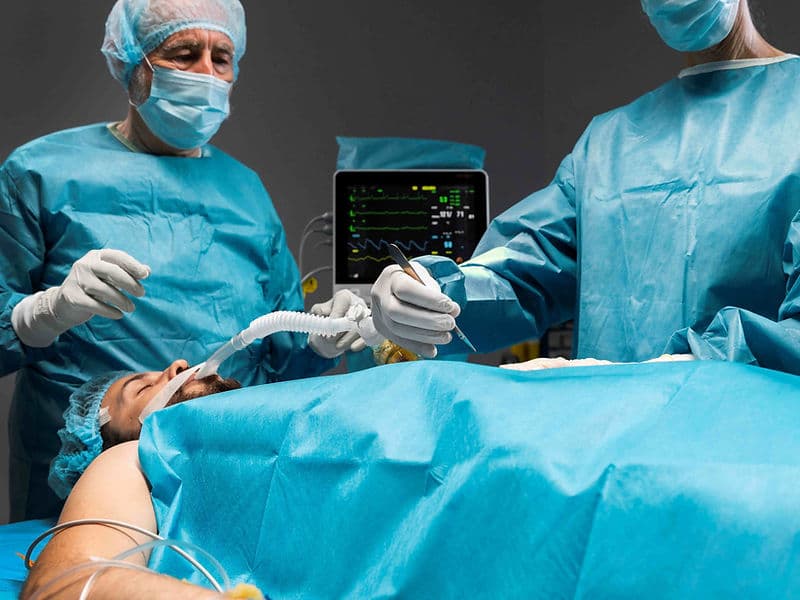
Next-Gen Life Support (ECMO & CPB): World-class heart-lung machines that can temporarily take over a patient's vital functions during surgery or recovery.
Advanced Monitoring Units (CCU): A specialized Coronary Care Unit where every heartbeat is tracked by a central "war room" for 24/7 safety.
Precision Wearable Diagnostics: Utilizing Holter monitors and wearable telemetry to catch intermittent rhythm issues over extended periods.
Comprehensive Diagnostic and Support Facilities
From arrival to recovery, every facility is designed to minimize risk and maximize surgical success:
Non-Invasive Stress Lab: Featuring TMT (Treadmill Test) and Stress-Echo facilities to evaluate the heart under physical exertion.
3D Cardiac Imaging Suite: Housing Cardiac MRI and CT Coronary Angiogram technology for detailed, scalpel-free arterial mapping.
Cardiac Biomarker Lab: On-site pathology units for rapid testing of heart-specific proteins (Troponin) during emergencies.
Rehabilitation & Wellness Wing: A dedicated space for monitored exercise programs and nutritional counseling for heart health.
International Patient Concierge: A private lounge where global families can coordinate medical documentation, insurance, and follow-up care.
A Commitment to Survival and Quality of Life
The department operates under a "Time is Muscle" philosophy, ensuring that patients receive the fastest and most precise care possible:
Rapid Reperfusion Protocols: Ensuring that blocked arteries are opened within the critical 90-minute window to prevent heart damage.
Valve Replacement Innovations: Offering minimally invasive TAVI/TAVR procedures to replace valves through a small tube, avoiding open-chest surgery.
Holistic Recovery Framework: Combining high-tech intervention with lifestyle-focused survivorship plans to ensure the heart remains healthy for decades.
Collaborative Decision-Making: Every complex case is reviewed by both a surgeon and a cardiologist to ensure the safest long-term outcome.
May we help you?
Related Treatments
Related Specialists

Dr Arul Dominic Furtado
Cardiothoracic & Vascular Surgeon (CTVS)
Aster CMI Hospital
15+years experience

Dr Anita Saxena
Pediatric Cardiac Surgeon
Fortis Escorts Heart Institute, New Delhi
40+years experience

Dr Nitin Kumar Sethi
Neurologist
Pushpawati Singhania Hospital & Research Institute
27+years experience