Cancer Care/Oncology
Oncology Department (Cancer Care)
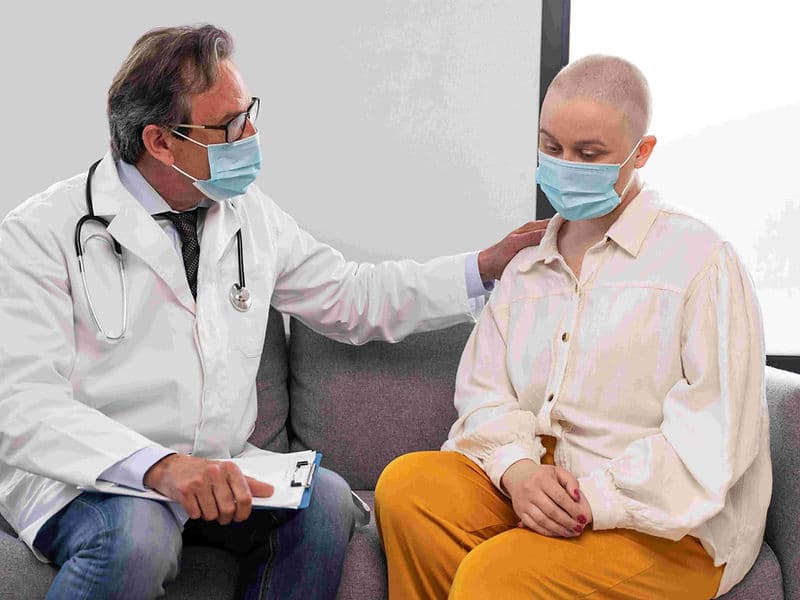
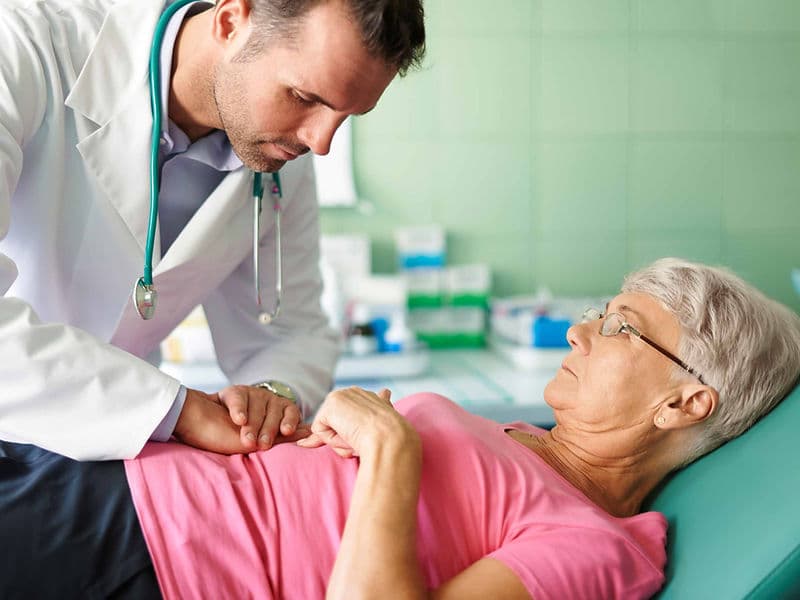
The Oncology Department is a premier center for comprehensive cancer care, designed as a sanctuary of hope and advanced healing. Modern oncology at this level is built upon a Multidisciplinary Team (MDT) framework, where every patient’s journey is overseen by a collective of experts. For international patients, this department represents the pinnacle of precision medicine, combining high-energy radiation, molecular systemic therapies, and surgical mastery to provide a holistic, 360-degree approach to recovery.
A Multidisciplinary Circle of Expertise
Effective cancer care requires a "Tumor Board" approach, where specialists collaborate to design a personalized treatment roadmap:
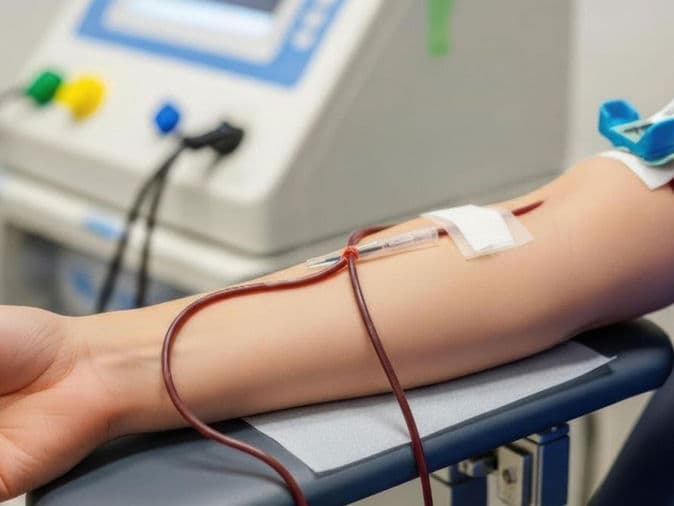
Medical Oncologists: The lead strategists who manage systemic treatments, including advanced Chemotherapy, Immunotherapy, and Targeted Therapy.
Surgical Oncologists: Specialists focused on the precision removal of tumors while preserving healthy surrounding tissue (margins).
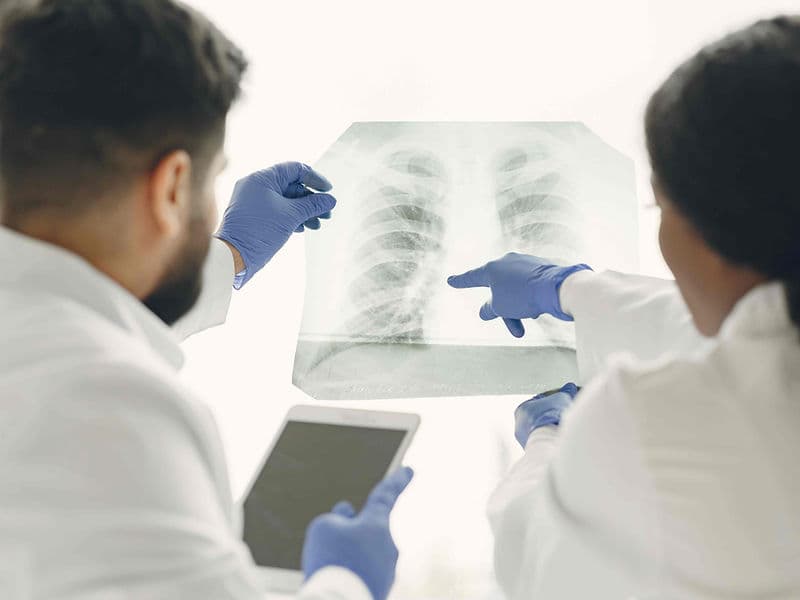
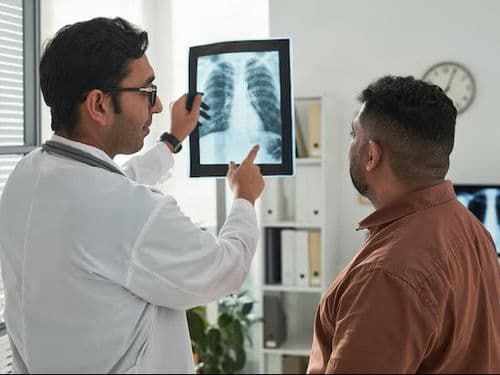
Radiation Oncologists: Experts who use high-energy subatomic particles to target and destroy cancer cells without the need for a scalpel.
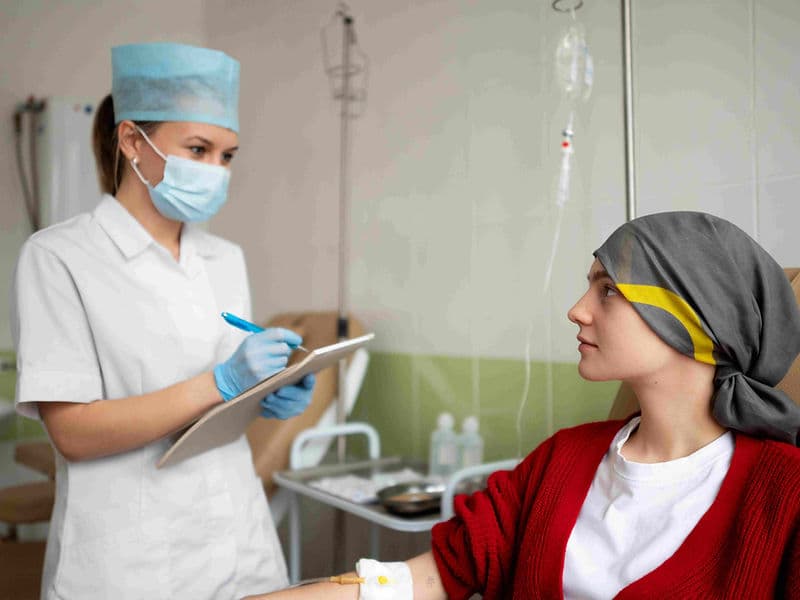
Haemato-Oncologists: Dedicated specialists for blood-based cancers, such as Leukemia and Lymphoma, often overseeing Bone Marrow Transplants.
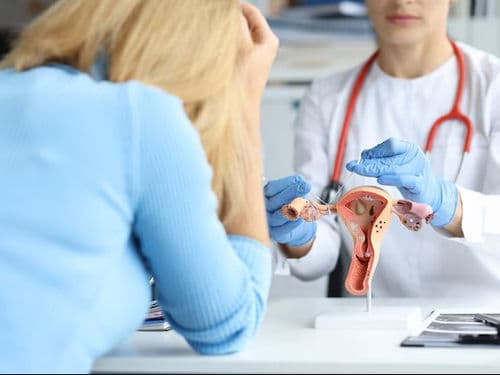
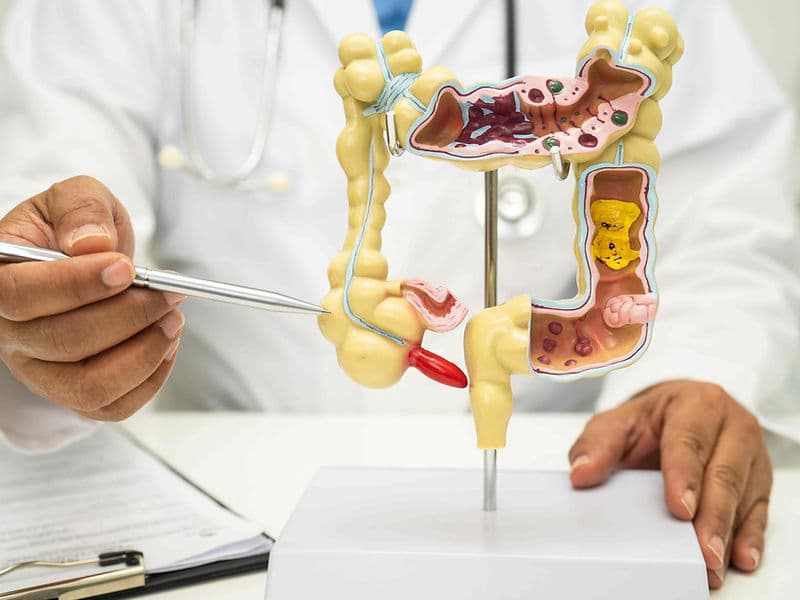
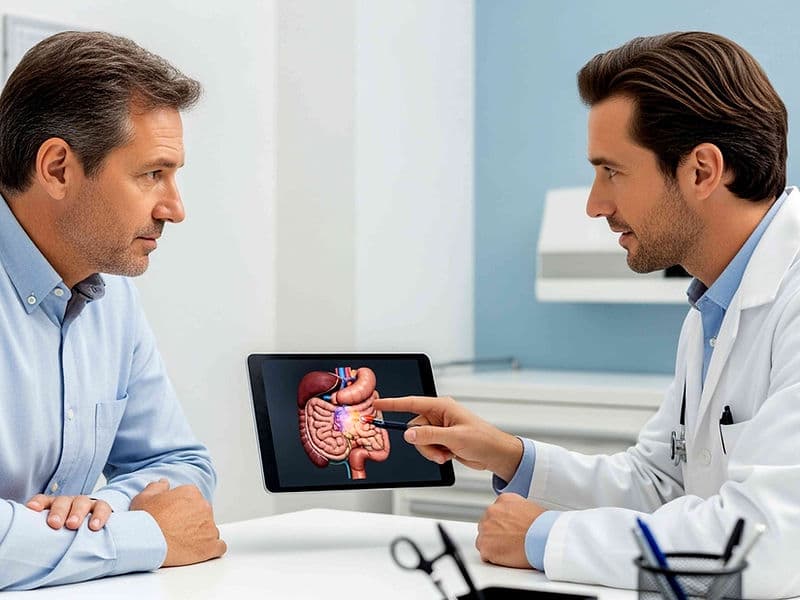
Onco-Pathologists: Vital researchers who decode the genetic signature of a tumor to identify the most effective drug matches.
Palliative Care Specialists: A compassionate team dedicated to symptom management and maintaining the highest possible quality of life.
Advanced Technology and Precision Tools
Our department utilizes the latest global innovations to ensure treatment is as effective as it is safe:
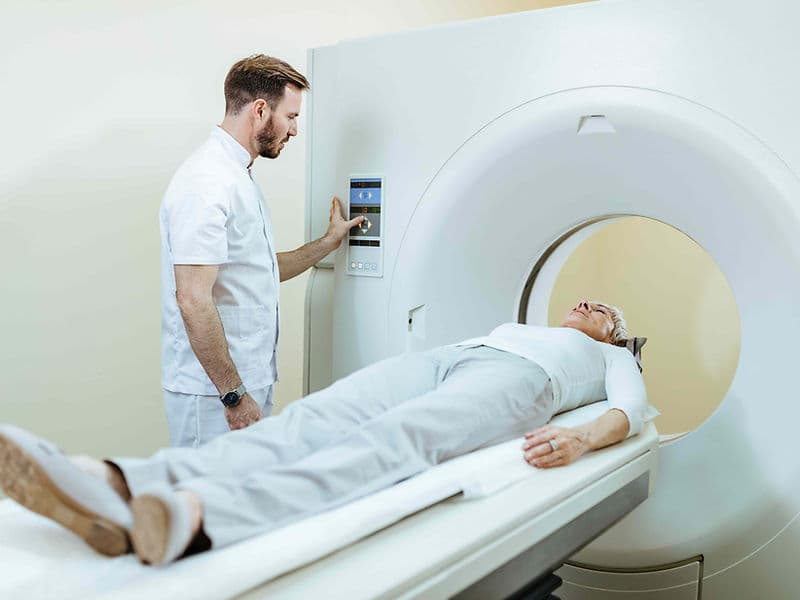
LINAC (TrueBeam) Technology: High-energy linear accelerators that synchronize with your breathing to target tumors with pinpoint accuracy, sparing healthy organs.
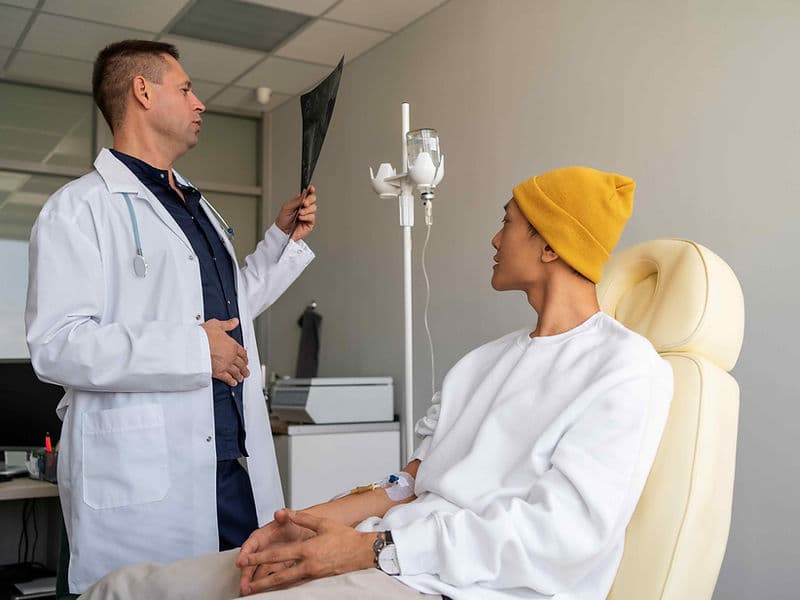
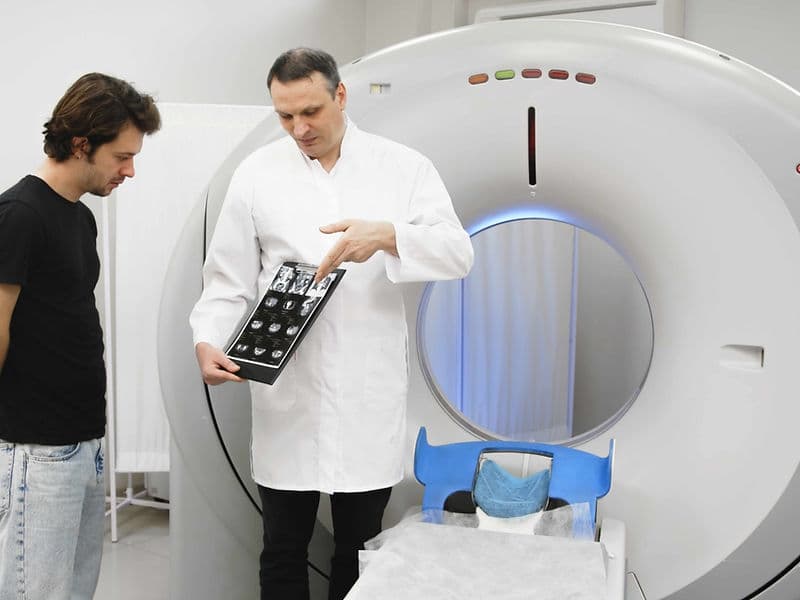
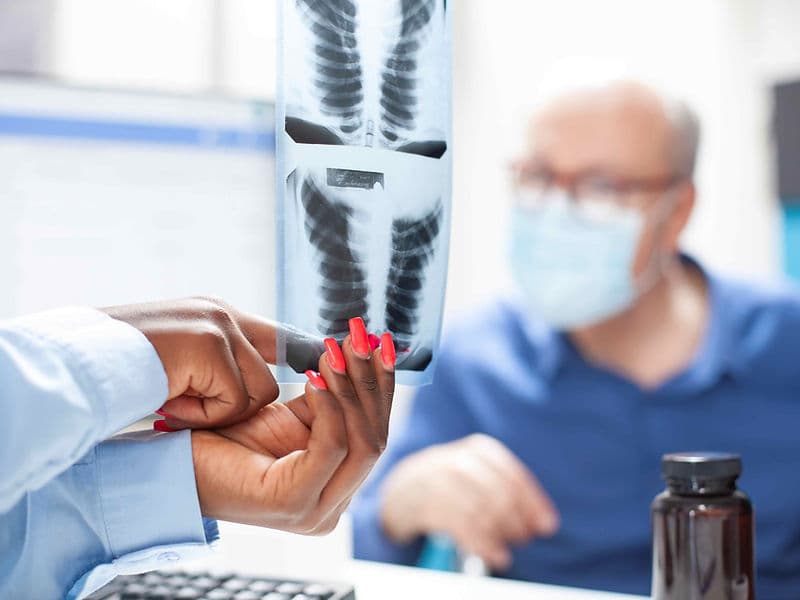
PET-CT Whole-Body Imaging: Advanced scanners that use radioactive markers to visualize active disease, helping to track treatment progress in real-time.
Molecular Targeted Therapy: Drugs designed to attack specific genetic mutations within cancer cells, sparing healthy cells and reducing side effects.
Scalpel-Free Radiosurgery: Non-invasive systems like Brachytherapy and localized radiation "seeds" that treat the tumor from the inside out.
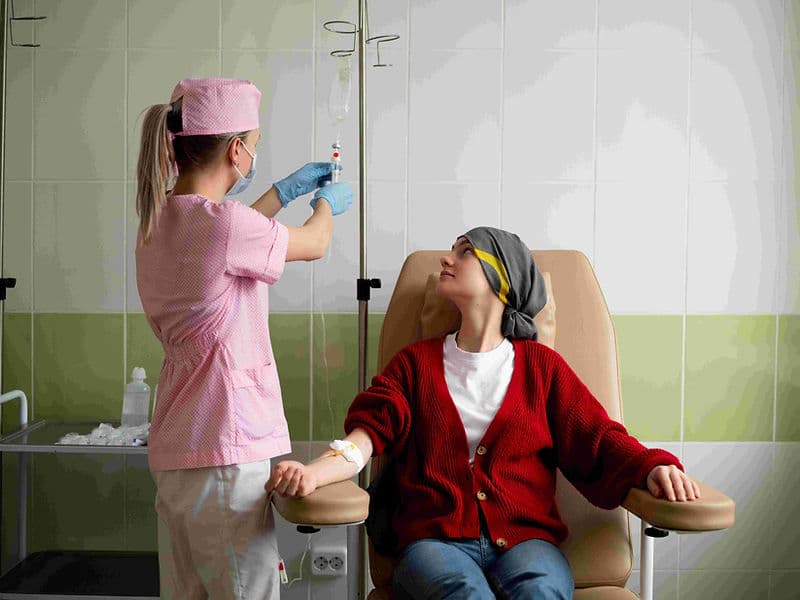
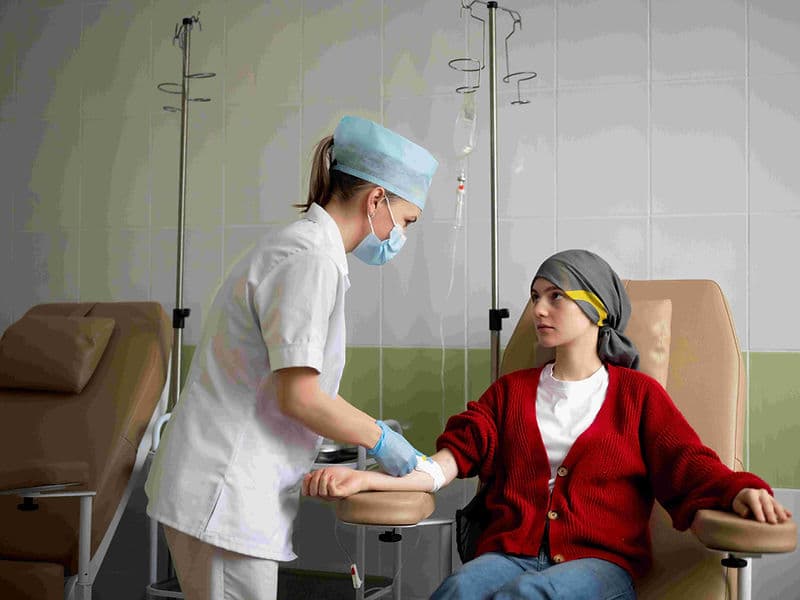
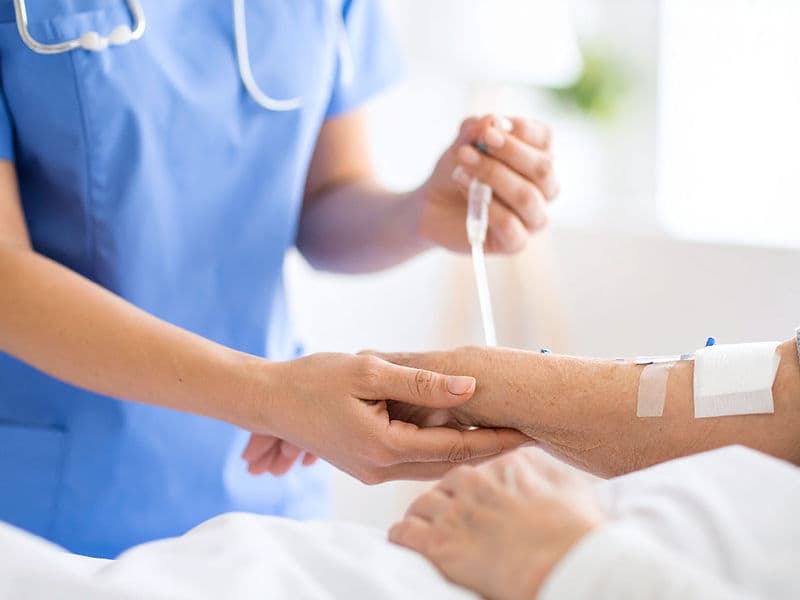
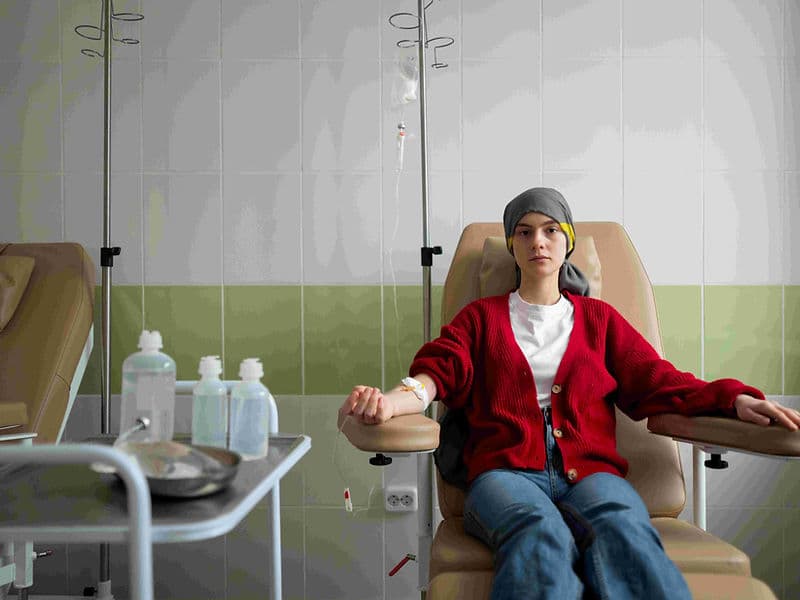
Digital Infusion Suites: High-tech "Daycare" centers equipped with precise infusion pumps and Cold Caps to minimize hair loss during therapy.
Specialized Care Units and Patient Facilities
To ensure safety and comfort, the department is organized into dedicated functional zones:
The Oncology Daycare Center: A premium, comfortable environment for outpatient chemotherapy infusions.
Bone Marrow Transplant (BMT) Unit: Ultra-sterile "Clean Rooms" with HEPA filtration systems designed for patients with sensitive immune systems.
The Tumor Board Room: A collaborative hub where surgeons, radiologists, and pathologists meet to review and refine individual patient cases.
Nuclear Medicine Wing: A lead-lined facility for specialized diagnostic scans and targeted isotope treatments like Radio-Iodine therapy.
International Patient Sanctuary: A dedicated lounge for global families to coordinate medical records, travel logistics, and long-term survivorship plans.
A Commitment to Long-Term Wellness
The care journey extends well beyond the final treatment session to ensure lasting health:
Survivorship Roadmaps: Comprehensive follow-up plans covering nutrition, surveillance scans, and exercise.
Maintenance Therapy: Ongoing access to the latest targeted pills to prevent recurrence and ensure long-term remission.
Psychosocial Support Groups: Integrated networks and counseling to help patients and families navigate the emotional transition to life after cancer.